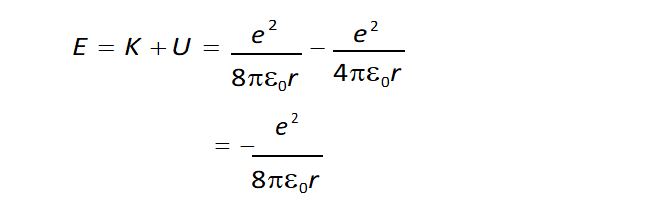Jagran Josh
Atoms Formulas & Diagrams: Students can find a list of important diagrams and formulas for CBSE Class 12 Physics Chapter 12, Atoms. Use the PDF download link attached below to save the formula sheet.
CBSE Class 12 Physics Atoms Formula list: The class 12 Physics CBSE Board Exam is one of the toughest board exams with the most critical question paper. A lot of effort and time goes into preparing for it. Since, all of us know that Physics is a subject full of formulas, derivations, graphs, and apt use of mathematical equations, it is important that all of these are combined and put together to save some time. Half of your time can be utilized if you have a sheet of formulas, diagrams, important definitions, graphs, etc which can be referred to, anytime.
To help you with the same, we have brought here the Formula sheet for CBSE Class 12 Physics Part 2, Chapter 2, Atoms. This formula sheet also consists of important diagrams that can be directly asked for in your CBSE Board Examination. If not directly, they can be given in the question paper to derive equations and formulas. For your convenience, we have also presented a PDF download link below. It is advised that students should use the link to download the formulas and use them while solving NCERT Solutions, in-text exercises, and solving numerical.
The class 12 Physics Atoms chapter talks about some concepts that can be important from the CBSE Board Exam’s point of view. Rutherford’s nuclear model of atom, Atomic spectra, Bohr model of the hydrogen atom, De Brogile’s explanation of Bohr’s Second postulate of quantization, and energy levels, are some of them.
Related Articles:
Formula Sheet for CBSE Class 12 Physics Chapter 12 Atoms
Diagrams
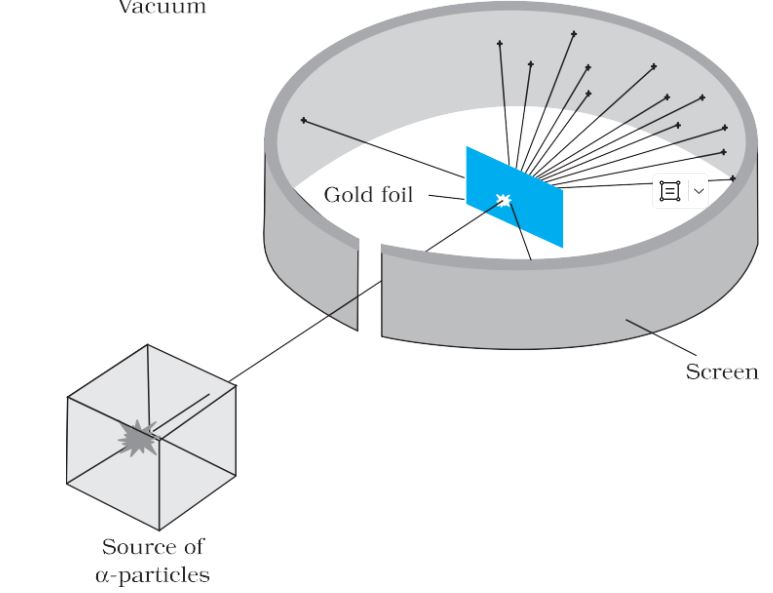
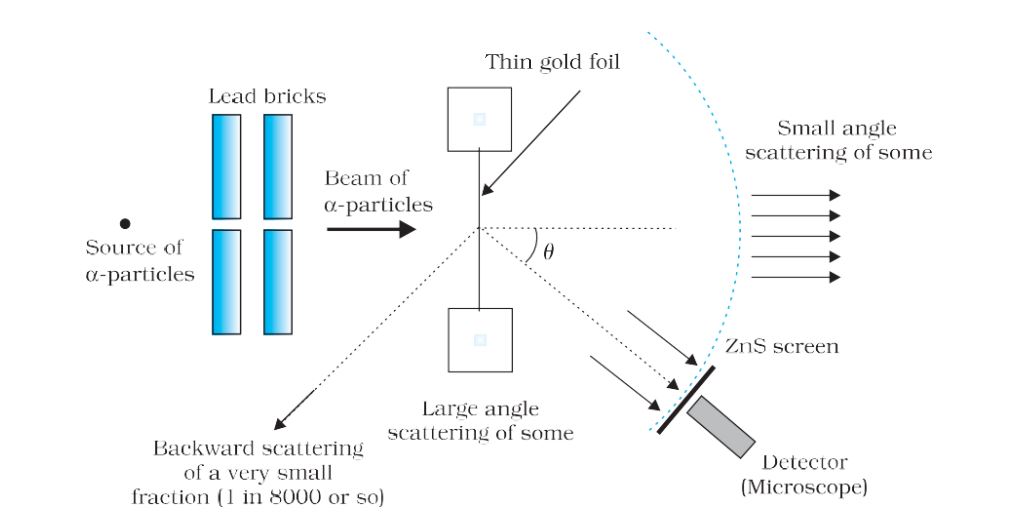
Geiger-Marsden scattering experiment
Schematic arrangement of Geiger-Marsden scattering experiment
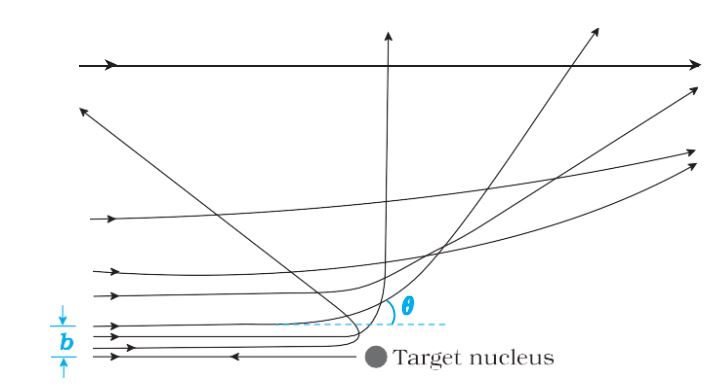
Alpha particle trajectory
Atomic Spectra: Emission lines in the spectrum of hydrogen
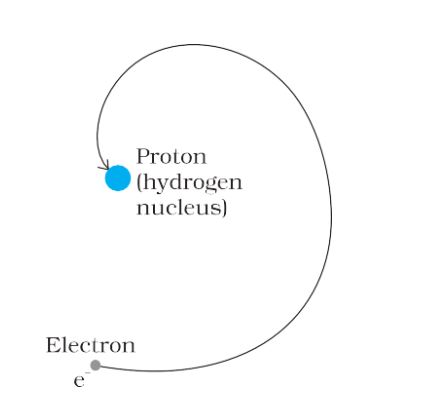
Bohr’s model of the hydrogen atom
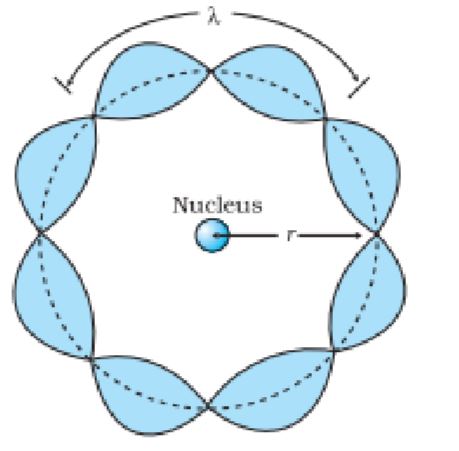
DE BROGLIE’S EXPLANATION OF BOHR’S SECOND POSTULATE OF QUANTISATION
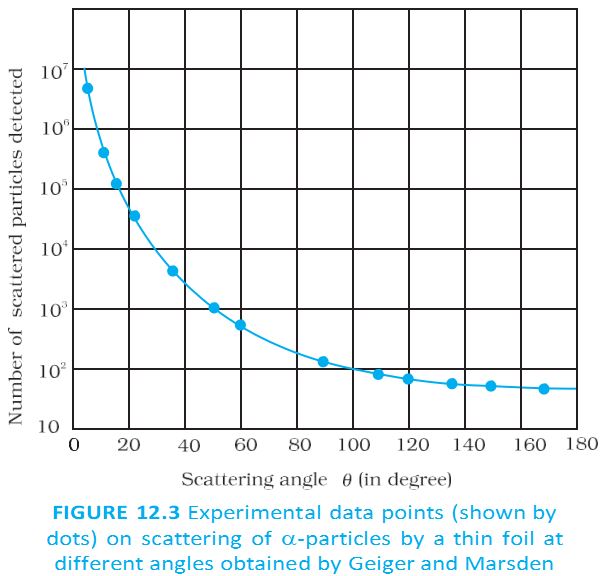
Scattering of alpha particles by a thin foil at different angles
Formulas:
Magnitude of force of α particle:
Conservation of Energy
Distance of closest approach
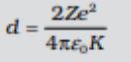
Total energy of the electron
Velocity of electron
Frequency of the electron moving around proton
Orbital Radius
Angular Momentum
Frequency of emitted photon
Radius of nth possible orbit
Energy of an electron
Difference in energy levels
Wavelength
Angular momentum of the electron(According to Bohr’s quantum condition)
To download formulas and diagrams for CBSE Class 12 Physics Chapter 12 Atoms are:
Also Find:
NCERT Solutions Class 12 Physics 2023-2024
#CBSE #Class #Physics #Atoms #Formula #List #Definitions #Diagrams